Technology
What is Atmospheric Plasma?
Plasma is the fourth state of matter, existing as a highly energetic gas with properties dependent on the chemical composition of the gas itself. In everyday life, people observe plasma in the aurorae, lightning, fluorescent light bulbs and use plasma through their spark plugs to start their car. Each of these applications have a plasma state that is different due to the gas composition, gas pressure, and the amount of energy applied. Plasma applications are typically separated by pressure (low, atmospheric, and high) and their thermodynamic properties (thermal and non-thermal). Examples of low-pressure plasma applications are semiconductor wafer cleaning and fluorescent light bulbs. High pressure plasmas are studied for supersonic combustion, fusion, and space reentry. In the past 30 years, atmospheric pressure plasmas have gained interest for their use in applications similar to low pressure applications, but without the expensive equipment required to maintain a low pressure environment.
Atmospheric pressure plasma is ignited at ambient temperature and pressure in a specific gas or gas mixtures such as air, carbon dioxide, argon, helium, water vapor, etc. The energy applied to these gases causes a “breakdown”, where gas molecules are ionized resulting in free electrons. These electrons then become energetic due to the energy applied and react with the surrounding gas to form reactive chemical species. At APS, we specialize in the creation of complete atmospheric plasma systems (plasma power supply and plasma generating head or applicator) to deliver the chemically activated plasma beam required for specific applications. Our flagship device, The PlasmaBlast PB7000 delivers a plasma beam to remove coatings, clean surfaces and promote adhesion with a single treatment.
How Atmospheric Plasma Works to Remove Coatings
The plasma generated by the PlasmaBlast 7000 series uses compressed air and electricity to generate a unique mixture of atomic and molecular reactive oxygen and nitrogen species. These species are delivered using the compressed gas to the surface of the coating (paint, primer, adhesive), where they interact with the carbon and hydrogen in the coating. The oxygen species will oxidize the backbone carbon-hydrogen bonds and weaken the binders in the coating. This results in the vaporization of the coating that can be seen when treating coatings with PlasmaBlast. The oxygen species that react with the carbon and hydrogen form carbon dioxide and water vapor respectively, as shown in the figure below. These gases are blown away from the surface along with the unused reactive species through the compressed airflow (90 slm). Organic coatings are removed layer by layer in this manner until the substrate underneath is revealed. The inorganics within the coating are oxidized, if possible, and released as a dry dust. The dust is then either blown off by the plasma treatment or is collected via a vacuum system if deemed potentially toxic due to the composition of the coating.
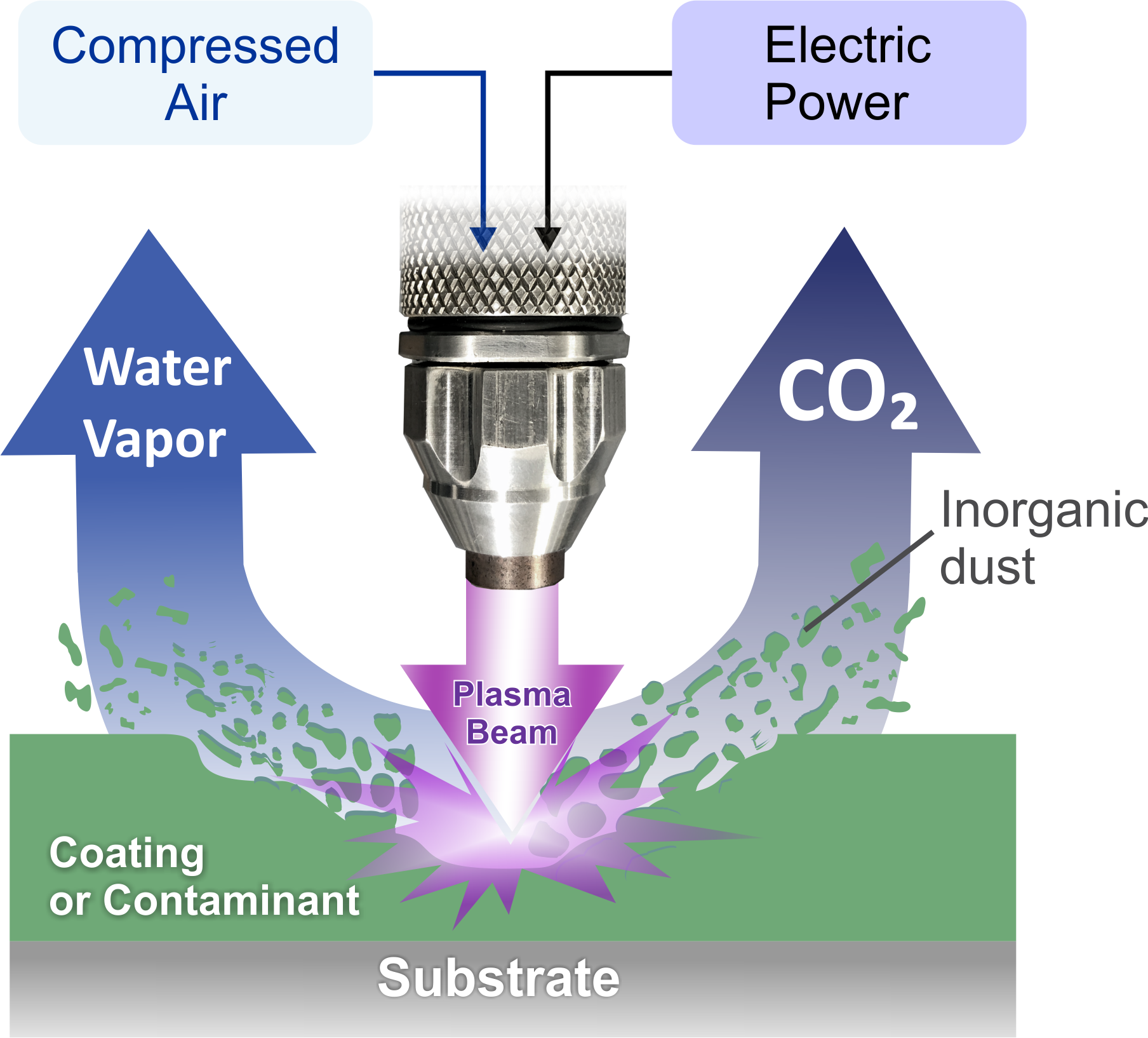
How It Cleans Surfaces
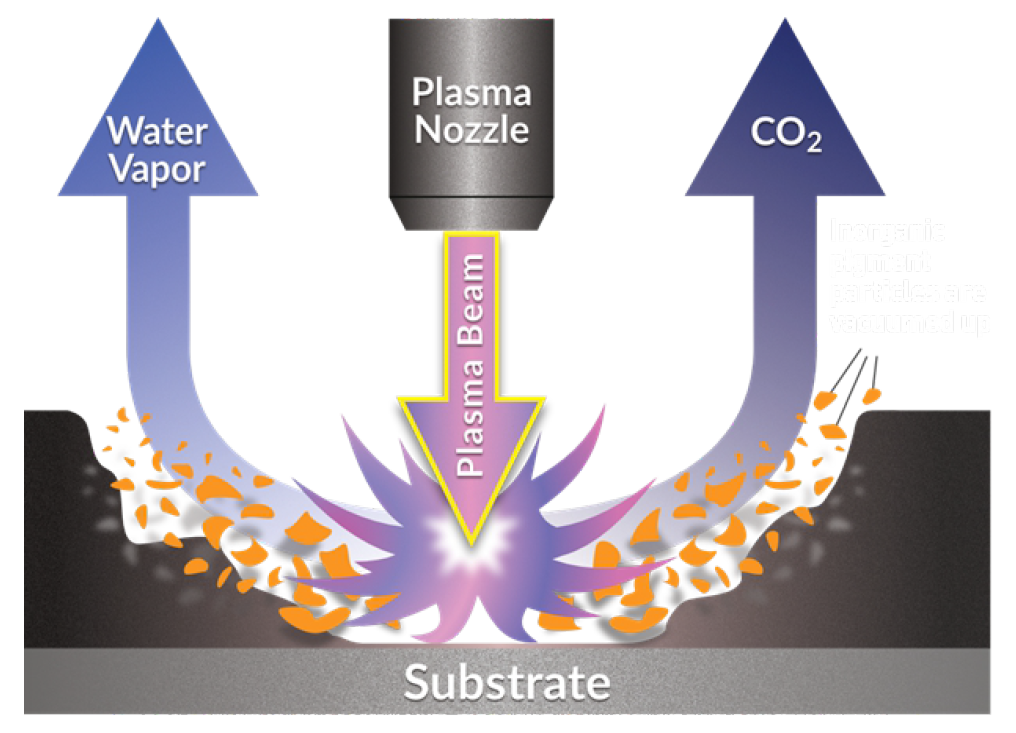
If the surface has coatings or contamination, the PlasmaBlast treatment will deliver the dry etching chemistry of the plasma beam to the surface where it will vaporize the contaminants, like oil or grease, and remove it. The gaseous products will then be swept away by the continuous gas flow of the treatment. This type of plasma treatment can replace most solvent-based or caustic, wet chemical processes to remove greases and oils prior to further processing of materials.
How It Promotes Adhesion
A clean surface itself will promote adhesion between the surface and a coating. This is due to increased contact, or surface area, at the interface between the surface and the coating. This interface layer is influenced by the presence of contaminants and the surface energy of the surface. The presence of contaminants will cause the coating to bond to the contaminant itself instead of the surface underneath which results in the paint being susceptible to failure at the spot. While most cleaning processes only remove contamination, plasma treatment also affects the surface energy. The addition of oxygen species to the bare surface causes the attachment of oxygen species and its intermediates (other chemical species containing oxygen) to the surface. These attachments replace the hydrogen bonds on the surface with oxygen bonds, which increases the surface energy. The increase in surface energy causes an increase in wettability, or how well water will wick over a surface. The more surface area the coating can cover i.e. the more it can wet out the surface, the better the bond between the coating and the surface. Therefore, the attachment of these oxygen species to the surface increases the density of chemical bonding sites between the coating and the surface and thus promotes adhesion. To confirm the surface is sufficiently treated, APS uses a special type of test equipment called a surface energy analyzer developed by BTG labs. This analyzer is used to measure the water contact angle of a surface, which provides an indirect measure of the surface energy. There is an inverse relationship between surface energy and water contact angles. Meaning a low water contact angle will indicate a high surface energy and good adhesive bonding. Typically surfaces with water contact angles less than 40° will exhibit good adhesive bonding. Depending on the plasma treatment time and the intensity, the PlasmaBlast system can produce water contact angles less than 10° which equates to extremely high surface area and superior adhesion.